Exploring the local electronic structure and geometric arrangement of ALD Zn(O,S) buffer layers using X-ray absorption spectroscopy†
Abstract
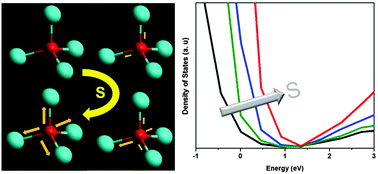
The growing interest in zinc oxysulfide (Zn(O,S)) thin films as buffer layers has been motivated by higher efficiencies achieved in solar cells. In this work we present insights into the electronic-geometric structure relationship of varying compositions of Zn(O,S) grown by atomic layer deposition (ALD). The X-ray absorption near edge structure (XANES), a local bonding-sensitive spectroscopic tool, with quantum simulations helps link the atomic structure to the unoccupied density of states (DOS) of the films. The infiltration of sulfur into a ZnO matrix results in the formation of S 3p–Zn 4sp–O 2p hybridized orbitals in the near edge X-ray absorption fine structure (NEXAFS) region of both the O and S K-edges. The extent of sulfur incorporation affects the ionicity of Zn, which in turn alters the bond lengths of Zn–O within the structure and its resulting bandgap. Knowing Zn(O,S)'s electronic-geometric structure interplay allows one to predict, tailor, and optimize its buffer layer performance.

 Please wait while we load your content...
Please wait while we load your content...