Effects of terminal chain length in hydrogen-bonded chiral switches on phototunable behavior of chiral nematic liquid crystals: helicity inversion and phase transition†
Abstract
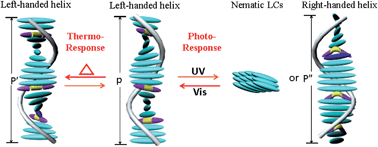
A novel series of photoresponsive chiral switches are fabricated by a facile hydrogen-bonded (H-bonded) assembly method, in which the binaphthyl azobenzene molecule is used as the proton acceptor, and binaphthyl acids with opposite chiral configuration are proton donors. We find that the helical twisted power of H-bonded chiral switches and the helical handedness of induced chiral nematic liquid crystals (N*-LCs) are mainly determined by the terminal flexible chain length in proton donors of binaphthyl acids. Controlling the lengths of the terminal flexible chain leads to different photoswitching behaviors by light irradiation, such as a helical inversion in the N*-LCs and a phase transition from N*-LCs to nematic LCs. This is mainly because of chiral counteraction and intensity attenuation of opposite chiral configurations between the proton acceptor and proton donor during UV-vis irradiation. Additionally, the thermal switching behavior of N*-LCs doped with H-bonded chiral switches is also demonstrated, and the related tuning mechanism may be attributed to the H-bonded effect and the changes in a dihedral angle of the binaphthyl rings. This facile assembly approach provides a new way for the fabrication of functional chiral switches for photonic applications.

 Please wait while we load your content...
Please wait while we load your content...