Catalytic dehydrogenative aromatization: an alternative route to functionalized arenes†
Abstract
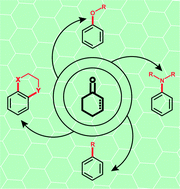
Catalytic dehydrogenative aromatization has emerged as an efficient and environmentally friendly way to access functionalized arenes in recent years. Typically, the compounds bearing an aliphatic six-membered ring, for instance, cyclohexanones, are used as arylation sources. The transformation process commonly involves a sequential, nucleophilic addition, dehydration and catalytically oxidative dehydrogenation, providing convenient entries to carbon–carbon and carbon–heteroatom bond formations. Compared to previous arylation methods, this strategy eliminates the use of harsh reaction conditions and the production of halide wastes, circumvents the issues of chem- and regioselectivities, and offers a milder/greener means for the synthesis of functionalized arenes. It is worth mentioning that this approach distinguishes itself from other methods by frequently using either oxygen as the sole oxidant or hydrogen transfer strategy. Furthermore, an obvious advantage, in terms of a sustainable chemical process, is that water is generated as the only by-product in most cases. This mini-review mainly focuses on our groups' contributions to the direct formation of C–O, C–N and C–C bonds using substituted cyclohexa(e)nones as flexible arylation sources and the extension of these methodologies to the construction of heterocycles.
- This article is part of the themed collections: HOT articles in Organic Chemistry Frontiers in 2015 and Celebrating the 80th Birthday of Professor Ei-ichi Negishi

 Please wait while we load your content...
Please wait while we load your content...