Polymerization of 5-alkyl δ-lactones catalyzed by diphenyl phosphate and their sequential organocatalytic polymerization with monosubstituted epoxides†
Abstract
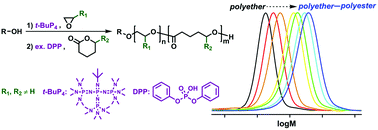
Organocatalytic ring-opening polymerization (ROP) reactions of three renewable 5-alkyl δ-lactones, namely δ-hexalactone (HL), δ-nonalactone (NL) and δ-decalactone (DL), using diphenyl phosphate (DPP) were investigated. Room temperature, together with a relatively high monomer concentration (≥3 M), was demonstrated to be suitable for achieving a living ROP behavior, a high conversion of the lactone, a controlled molecular weight and a low dispersity of the polyester. HL, containing a 5-methyl substituent, showed a much higher reactivity (polymerization rate) and a slightly higher equilibrium conversion than the compounds with longer alkyl substituents (NL and DL). The effectiveness of DPP-catalyzed ROP of 5-alkyl δ-lactones facilitated the one-pot performance following the t-BuP4-promoted ROP of monosubstituted epoxides. It has been shown in an earlier study that substituted polyethers acted as “slow initiators” for non-substituted lactones. However, efficient initiations were observed in the present study as substituted lactones were polymerized from the substituted polyethers. Therefore, this reinforces the previously developed “catalyst switch” strategy, making it a more versatile tool for the synthesis of well-defined polyether–polyester block copolymers from a large variety of epoxide and lactone monomers.
- This article is part of the themed collection: Celebrating The Five-Year Anniversary of Polymer Chemistry

 Please wait while we load your content...
Please wait while we load your content...