Iodine promoted α-hydroxylation of ketones†
Abstract
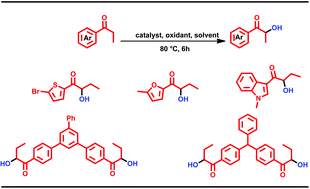
A novel method for α-hydroxylation of ketones using substoichiometric amount of iodine under metal-free conditions is described. This method has been successfully employed in synthesizing a variety of heterocyclic compounds, which are useful precursors. α-Hydroxylation of diketones and triketones are illustrated. This strategy provides a novel, efficient, mild and inexpensive method for α-hydroxylation of aryl ketones using a sub-stoichiometric amount of molecular iodine.

 Please wait while we load your content...
Please wait while we load your content...