Conformational properties of 1,4- and 1,5-substituted 1,2,3-triazole amino acids – building units for peptidic foldamers†
Abstract
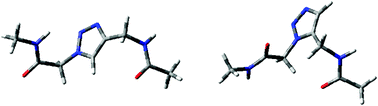
Peptidic foldamers have recently emerged as a novel class of artificial oligomers with properties and structural diversity similar to that of natural peptides, but possessing additional interesting features granting them great potential for applications in fields from nanotechnology to pharmaceuticals. Among these, foldamers containing 1,4- and 1,5-substitued triazole amino acids are easily prepared via the Cu- and Ru-catalyzed click reactions and may offer increased side chain variation, but their structural capabilities have not yet been widely explored. We here describe a systematic analysis of the conformational space of the two most important basic units, the 1,4-substitued (4Tzl) and the 1,5-substitued (5Tzl) 1,2,3-triazole amino acids, using quantum chemical calculations and NMR spectroscopy. Possible conformations of the two triazoles were scanned and their potential minima were located using several theoretical approaches (B3LYP/6-311++G(2d,2p), ωB97X-D/6-311++G(2d,2p), M06-2X/6-311++G(2d,2p) and MP2/6-311++G(2d,2p)) in different solvents. BOC-protected versions of 4Tzl and 5Tzl were also prepared via one step transformations and analyzed by 2D NOESY NMR. Theoretical results show 9 conformers for 5Tzl derivatives with relative energies lying close to each other, which may lead to a great structural diversity. NMR analysis also indicates that conformers preferring turn, helix and zig-zag secondary structures may coexist in solution. In contrast, 4Tzl has a much lower number of conformers, only 4, and these lack strong intraresidual interactions. This is again supported by NMR suggesting the presence of both extended and bent conformers. The structural information provided on these building units could be employed in future design of triazole foldamers.

 Please wait while we load your content...
Please wait while we load your content...