Rare earth 3-(4′-hydroxyphenyl)propionate complexes†
Abstract
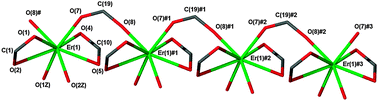
The reaction of lanthanoid chlorides or nitrates with sodium 3-(4′-hydroxyphenyl)propionate (Na4hpp) in methanol or water has yielded complexes [La4(4hpp)12(H2O)6]·4H2O·MeOH (1), [Ce2(4hpp)6(H2O)3]·(H2O)·2.5(EtOH) (2a) (after crystallization from ethanol), [Ho(4hpp)3(H2O)2] (5), [Er(4hpp)3(H2O)2]·1.5(H2O) (6), and [Lu(4hpp)3]·H2O crystal composition (7), as well as heterobimetallics [NaCe2(4hpp)7(H2O)2]·3(H2O) (2b), [NaPr2(4hpp)7(H2O)2]·3(H2O) (3), and [NaNd2(4hpp)7(H2O)(MeOH)]·(H2O)·3(MeOH) (4). The structures of homometallic complexes 1, 2a, 6, and 7 reveal one-dimensional coordination polymers and vividly illustrate the effect of lanthanoid contraction with a decline in coordination numbers in the series from 9–11 (1), 9,10 (2a), 8 (6) to 7 (7) through variations in carboxylate coordination and ligation of water. Bimetallic complexes 2a and 4 each exhibit five different carboxylate binding modes as well as coordination of the 4-OH substituent of 4hpp to sodium thereby linking 1D polymer chains into a 2D network with both 9 and 10 coordinate Ln atoms and 6 coordinate sodium. Bulk products after drying lose solvent of crystallization in some cases (2a, 6), or exchange MeOH for water (4). X-ray powder diffraction indicates that bulk 2b and 3 are isotypic, as are bulk 5 and 6. In contrast to the excellent corrosion protection of lanthanum 4-hydroxycinnamate, compound 1 is ineffective in preventing the corrosion of mild steel, thereby establishing the importance of the –CH![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) CH– structural unit of the former in its anti-corrosion properties. However the flexible –CH2–CH2– chain of the 4hpp ligand enables the crystal engineering of its lanthanoid complexes in a wide variety of structures as well as effective crystallization for structure determination, whereas the analogous 4-hydroxycinnamates have so far evaded structural characterization except for Ln = La, Ce owing to crystallization problems.
CH– structural unit of the former in its anti-corrosion properties. However the flexible –CH2–CH2– chain of the 4hpp ligand enables the crystal engineering of its lanthanoid complexes in a wide variety of structures as well as effective crystallization for structure determination, whereas the analogous 4-hydroxycinnamates have so far evaded structural characterization except for Ln = La, Ce owing to crystallization problems.
- This article is part of the themed collection: Frontiers of Organo-f-Element Chemistry

 Please wait while we load your content...
Please wait while we load your content...