DFT/TDDFT investigation on the UV-vis absorption and fluorescence properties of alizarin dye†
Abstract
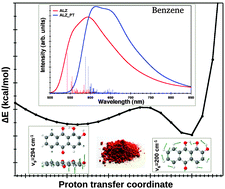
The photophysical and photochemical properties of alizarin, a fluorescent organic red dye of the family of the anthraquinones, have been theoretically investigated by focusing our attention on its emission properties in relation to an excited-state internal proton transfers from the phenolic hydroxyl group to the carbonyl oxygen. The potential energy curve of the proton transfer in the first excited state has been computed in solvents of different polarity and the emission spectra of both tautomers simulated, including the vibronic effects, using the Franck–Condon approximation. Calculations performed by equilibrating the solvent with the excited-state geometry and electron density using a self-consistent procedure have led to interesting differences with respect to their linear response counterpart. The results obtained point out that, while the emission energy of alizarin is sensitive to solvent polarity, that of the proton-transfer tautomer is computed at similar wavelengths independently of the solvent. Comparison between computed and experimental data has allowed us to rationalize the alizarin double emission measured in non-polar solvents.

 Please wait while we load your content...
Please wait while we load your content...