Preparation of Co3O4 electrode materials with different microstructures via pseudomorphic conversion of Co-based metal–organic frameworks†
Abstract
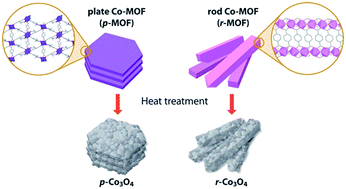
To develop high-performance nanostructured metal oxide electrodes, it is important to understand their structural effects on electrochemical performances. Thus, the preparation of metal oxide materials that have well-tailored nanostructures is crucial for studies. However, while synthetic strategies to control the size of metal oxide nanoparticles are well-developed, the control of the higher level structures, namely microstructure, is not very well established. Herein, we present the synthesis of the two kinds of Co3O4 nanomaterials through pseudomorphic conversion so that the macroscopic morphologies of parent MOFs, such as plate-like and rod-like shape, are well-maintained. Both Co3O4 nanomaterials are composed of almost identical 10 nm-sized primary nanocrystals but with different nanoporous secondary structures and macroscopic morphologies such as plate and rod shapes. These Co3O4 nanomaterials were utilized as an electrode in lithium ion batteries (LIBs), and their electrochemical properties were comparatively investigated. It was revealed that the different cyclability and rate capability are attributed to their different microstructures. The pseudo-monolithic integration of primary and secondary structures at higher level was the governing factor, which determined the electrochemical performances of the Co3O4 electrode.

 Please wait while we load your content...
Please wait while we load your content...