Proton conductivity enhancement in oriented, sulfonated polyimide thin films†
Abstract
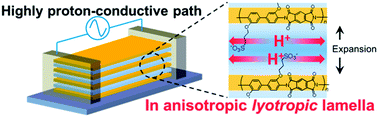
Studies of proton transport in confined thin polymer electrolytes are essential for providing additional information regarding the structure–property relationships of such materials. Using a combination of proton transport measurements and structural characterization, we explored the effect of proton conductivity in sulfonated polyimide (SPI) under both bulk and nanostructured thin film systems. A SPI film confined to a thickness of approximately 530 nm shows significant proton conductivity enhancement to a value of 2.6 × 10−1 S cm−1 (95% RH at 298 K), which is almost one order of magnitude more proton conductive than the bulk system (3.0 × 10−2 S cm−1 at 90% RH and 298 K). In thin films, the preferred chain packing along the in-plane direction can have considerable influence on the charge transport characteristics, which leads to the enhanced proton conductivity. Infrared (IR) p-polarized multiple-angle incidence resolution spectrometry (p-MAIRS) and in situ grazing-incidence small-angle X-ray scattering (GISAXS) were used to investigate the direction of polymer orientation and the changes in the internal polymeric structure under various humidity conditions, respectively. Under high-humidity conditions, the strong interaction between the side-chain sulfonic acid groups and water molecules causes an abrupt change in the internal structure in bulk SPI. Such a structural rearrangement results in a liquid-crystal-like ordered polymer structure. The RH dependent FTIR-ATR studies reveal that the accumulation of a large fraction of water molecules with stronger hydrogen bonding at high humidity regions causes the huge deviation in the internal morphology of the bulk samples.

 Please wait while we load your content...
Please wait while we load your content...