Understanding the pathway of antibacterial activity of copper oxide nanoparticles†
Abstract
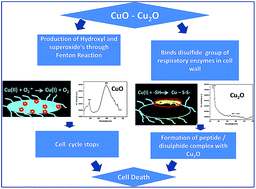
This work investigates the role of oxidation state in the antibacterial activity of copper oxide nanoparticles (NPs). The findings add strong support to a contact killing mechanism of copper oxides (CuO and Cu2O) through which bacteria initially suffer severe damage to the cell envelope. Then further damage ensues by an independent pathway of each copper oxide nanoparticle. Formation of copper(I)–peptide complex from cuprous oxide (Cu2O) and free radical generation from cupric oxide (CuO) were identified as key sources of toxicity towards E.coli. Cu2O rapidly inactivated Fumarase A, an iron sulphur cluster enzyme suggesting the cuprous state of copper binding to the proteins. This inactivation was not noticed in CuO. The percentage of biocidal/bacteriostatic activity is closely related to the oxidation state of the copper oxides. In the case of E.coli, Cu2O nanoparticles showed more efficient antibacterial activity and higher affinity to the bacterial cells. CuO nanoparticles produced significant ROS in terms of super oxides while Cu2O did not. The diminishing defective emission peaks of Cu2O after incubation with microbes strongly suggest the formation of protein complexes. This work is carried out to enable better understanding of the mechanistic pathways of copper oxide nanoparticles.
- This article is part of the themed collection: Chemistry in the battle against infections

 Please wait while we load your content...
Please wait while we load your content...