Accuracy of the analysis of free vancomycin concentration by ultrafiltration in various disease states
Abstract
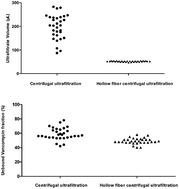
In the present study, the accuracy of the analysis of free vancomycin (VCM) by ultrafiltration in various disease conditions was assessed. With VCM as a representative drug, we used clinical plasma samples to investigate the effect of plasma conditions on the volume ratio of ultrafiltrate to sample solution (Vu/Vs) and the consequential effect on the measured free drug concentration (fc). Our results demonstrated that plasma conditions had a significant impact on Vu/Vs by centrifugal ultrafiltration (CF-UF). The Vu by CF-UF ranged from 97 μL to 279 μL among different individuals under the same centrifugation conditions. Total protein levels and the osmotic pressure of plasma were the main influence factors of Vu/Vs in disease states. In contrast, the Vu/Vs by hollow fiber centrifugal ultrafiltration (HFCF-UF) was less influenced by plasma conditions. As a consequence, the results of fc determined by HFCF-UF were more accurate than those by CF-UF for patients with different disease states. HFCF-UF displayed great advantages in clinical samples for accurate analysis of fc. It has been successfully applied to monitor free VCM in clinical plasma samples in routine therapeutic drug monitoring (TDM).

 Please wait while we load your content...
Please wait while we load your content...