Deproto-metallation using mixed lithium–zinc and lithium–copper bases and computed CH acidity of 2-substituted quinolines†
Abstract
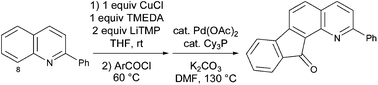
2-Substituted quinolines were synthesized, and their deproto-metallation using the bases prepared by mixing LiTMP with either ZnCl2·TMEDA (1/3 equiv.) or CuCl (1/2 equiv.) was studied. With phenyl and 2-naphthyl substituents, the reaction occurred at the 8 position of the quinoline ring, affording the corresponding iodo derivatives or 2-chlorophenyl ketones using the lithium–zinc or the lithium–copper combination, respectively. With a 4-anisyl substituent, a dideprotonation at the 8 and 3′ position was noted using the lithium–zinc base. With 3-pyridyl, 2-furyl and 2-thienyl substituents, the reaction took place on the substituent, at a position adjacent to its heteroatom. 2-Chlorophenyl-2-phenyl-8-quinolyl ketone could be cyclized under palladium catalysis. The experimental results were analyzed with the help of the CH acidities of the substrates, determined in THF solution using the DFT B3LYP method.

 Please wait while we load your content...
Please wait while we load your content...