Design, synthesis and molecular docking of novel bipyrazolyl thiazolone scaffold as a new class of antibacterial agents†
Abstract
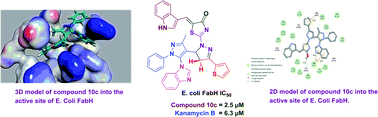
A new series of bipyrazolyl thiazolone hybrids were designed based on molecular hybridization technique. All the synthesized compounds were characterized by elemental analysis and various spectroscopic methods. They were tested for their in vitro antibacterial activity against two Gram-positive and two Gram-negative bacteria as well as E. coli FabH using Kanamycin B and Penicillin G as the standard drugs. Of the compounds studied, compound 10c (IC50 = 2.1 μM) showed the most effective E. coli FabH inhibitory activity as compared to other members of the series. The molecular docking study indicated that compound 10c was found nicely bound into the active site of E. coli FabH with hydrogen bonds, π–π and π–cation interactions having minimum binding energy ΔGb = −52.27 kcal mol−1.

 Please wait while we load your content...
Please wait while we load your content...