Investigating the functional implications of reinforcing feedback loops in transcriptional regulatory networks†
Abstract
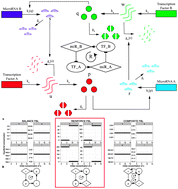
Transcription factors (TFs) and microRNAs (miRNAs) can jointly regulate transcriptional networks in the form of recurrent circuits or motifs. A motif can be divided into a feedforward loop (FFL) and a feedback loop (FBL). Incoherent FFLs have been the recent focus due to their potential to dampen gene expression noise in maintaining physiological norms. However, a cell is not only able to manage noise but also able to exploit it during development or tumorigenesis to initiate radical transformation such as cell differentiation or metastasis. A plausible mechanism may involve reinforcing FBLs (rFBLs), which amplify changes to a sufficient level in order to complete the state transition. To study the behaviour of rFBLs, we developed a novel theoretical framework based on biochemical kinetics. The proposed rFBL follows a parsimonious design, involving two TFs and two miRNAs. A simulation study based on our model suggested that a system with rFBLs is robust to only a certain level of fluctuation but prone to a complete paradigm shift when the change exceeds a threshold level. To investigate the natural occurrence of rFBLs, we performed a rigorous network motif analysis using a recently available TF/miRNA regulatory network from the Encyclopedia of DNA Elements (ENCODE). Our analysis suggested that the rFBL is significantly depleted in the observed network. Nonetheless, we identified 9 rFBL instances. Among them, we found a double-rFBL involving three TFs SUZ12/BCLAF1/ZBTB33 and three miRNAs miR-9/19a/129-5p, which together serve as an intriguing toggle switch between nerve development and telomere maintenance. Additionally, we investigated the interactions implicated in the rFBLs using expression profiles of cancer patients from The Cancer Genome Atlas (TCGA). Together, we provided a novel and comprehensive view of the profound impacts of rFBLs and highlighted several TFs and miRNAs as the leverage points for potential therapeutic targets in cancers due to their eminent roles in the identified rFBLs.

 Please wait while we load your content...
Please wait while we load your content...