Individually addressable arrays of replica microbial cultures enabled by splitting SlipChips†
Abstract
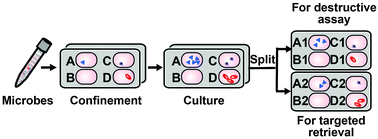
Isolating microbes carrying genes of interest from environmental samples is important for applications in biology and medicine. However, this involves the use of genetic assays that often require lysis of microbial cells, which is not compatible with the goal of obtaining live cells for isolation and culture. This paper describes the design, fabrication, biological validation, and underlying physics of a microfluidic SlipChip device that addresses this challenge. The device is composed of two conjoined plates containing 1000 microcompartments, each comprising two juxtaposed wells, one on each opposing plate. Single microbial cells are stochastically confined and subsequently cultured within the microcompartments. Then, we split each microcompartment into two replica droplets, both containing microbial culture, and then controllably separate the two plates while retaining each droplet within each well. We experimentally describe the droplet retention as a function of capillary pressure, viscous pressure, and viscosity of the aqueous phase. Within each pair of replicas, one can be used for genetic analysis, and the other preserves live cells for growth. This microfluidic approach provides a facile way to cultivate anaerobes from complex communities. We validate this method by targeting, isolating, and culturing Bacteroides vulgatus, a core gut anaerobe, from a clinical sample. To date, this methodology has enabled isolation of a novel microbial taxon, representing a new genus. This approach could also be extended to the study of other microorganisms and even mammalian systems, and may enable targeted retrieval of solutions in applications including digital PCR, sequencing, single cell analysis, and protein crystallization.

 Please wait while we load your content...
Please wait while we load your content...