Simultaneous and selective recovery of cellulose and hemicellulose fractions from wheat bran by supercritical water hydrolysis†
Abstract
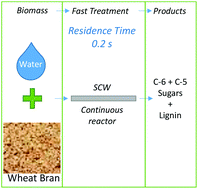
Supercritical water (SCW) has been demonstrated to be an excellent solvent and reaction medium to improve the cellulose hydrolysis selectivity by controlling the reaction time. In this study the conversion of wheat bran into soluble saccharides such as glucose, xylose and arabinose was analysed at 400 °C and 25 MPa with reaction times between 0.2 and 1 s. The process yield was evaluated for two different products: C-6 (glucose derived from cellulose) and C-5 sugars (saccharide derived from hemicellulose hydrolysis). The production of glycolaldehyde, furfural and 5-hydroxymethylfural (5-HMF) was analysed as by-product formation. Operation under supercritical conditions allows a biomass liquefaction of 84% w/w at 0.3 s of residence time. The obtained solid after the hydrolysis was composed of mainly lignin (86% w/w). The highest recovery of cellulose (C-6) and hemicellulose (C-5) as soluble sugars (73% w/w) was achieved at 0.19 s of reaction time. An increase in the reaction time decreased the yield of C-6 and C-5. A total recovery of C-5 was achieved at 0.19 s. On the other hand, the highest yield (65% w/w) of C-6 was achieved at 0.22 s of reaction time. The main hydrolysis product of C-6 and C-5 was glycolaldehyde, yielding 20% w/w at 0.22 s of reaction time. Furfural and 5-HMF production was highly inhibited under the experimental conditions, obtaining yields lower than 0.5% w/w. The hydrolysis reactions were performed in a continuous pilot plant at 400 °C, 25 MPa and residence times between 0.1 s and 0.7 s.

 Please wait while we load your content...
Please wait while we load your content...