Unprecedented dinuclear Robson type macrocyclic complexes having two +iii metal ions in two compartments and the role of the diimino moiety on the stability of metal ion oxidation states†
Abstract
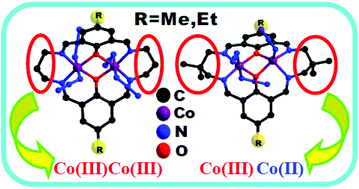
The work in this investigation deals with the syntheses, characterization, crystal structures and catechol oxidase activity of four diphenoxo-bridged CoIIICoIII compounds of composition [CoIIICoIIILMe–pn(N3)4]·6H2O (1), [CoIIICoIIILMe–pn(N3)4]·4H2O (2), [CoIIICoIIILEt–pn(N3)4]·3.5H2O (3) and [CoIIICoIIILEt–pn(N3)4]·CH3CN·2.5H2O (4), and two diphenoxo-bridged CoIIICoII compounds of composition [CoIIICoIILMe–Me2pn(N3)3]·2H2O (5) and [CoIIICoIILEt–Me2pn(N3)3]·H2O (6). In these compounds, H2LMe–pn, H2LEt–pn, H2LMe–Me2pn and H2LEt–Me2pn are four Robson type tetraiminodiphenolate macrocyclic ligands, in which the superscript Me and Et indicate that the dialdehyde components are 4-methyl-2,6-diformyl-phenol and 4-ethyl-2,6-diformyl-phenol, respectively, while pn and Me2pn indicate that the diamine components are 1,3-diaminopropane and 2,2-dimethyl-1,3-diaminopropane, respectively. The CoIIICoIII compounds 1–4 are further characterized by 1H NMR spectra. Compounds 1 and 3 are prepared following a metal-templated synthesis, while compounds 2 and 4 are prepared by direct synthesis, which includes using a pre-isolated macrocycle, [H4LMe–pn](ClO4)2/[H4LEt–pn](ClO4)2, as a reactant. On changing the diamine component of the corresponding macrocycle from 1,3-diaminopropane to 2,2-dimethyl-1,3-diaminopropane, but otherwise following the similar reaction procedure (both direct and template), the CoIIICoII compounds [CoIIICoIILMe–Me2pn(N3)3]·2H2O (5), [CoIIICoIILEt–Me2pn(N3)3]·H2O (6), [CoIIICoIILMe–Me2pn(N3)3]·0.5MeCN·0.27H2O (5A; Inorg. Chim. Acta, 2014, 412, 38) and [CoIIICoIILEt–Me2pn(N3)3]·MeCN (6A; Dalton Trans., 2013, 42, 4561) are formed, rather than CoIIICoIII compounds. Like compounds 5A and 6A, all the compounds 1–6 show catechol oxidase activity (substrate: 3,5-di-tert-butyl catechol). The kinetic parameters for 1, 3 and 6 have been determined. Their Kcat values are, respectively, 212.6, 188.0 and 191.3 h−1. ESI-MS (positive) spectra of some representative cases (with and without the substrate) have been recorded and the positive species have been assigned well. Unprecedented and surprising aspects of the composition/structure and properties of the complexes in this investigation have been discussed.

 Please wait while we load your content...
Please wait while we load your content...