N-2-Hydroxybenzaldehyde acylhydrazone–Fe(iii) complex: synthesis, crystal structure and its efficient and selective N-methylation†
Abstract
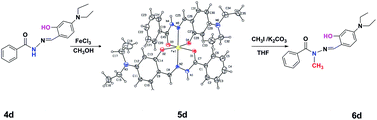
N-Methyl-N′-2-hydroxybenzaldehyde acylhydrazones have been chemospecifically synthesized in good yield by N-methylation of the Fe(III) complexes of N-2-hydroxybenzaldehyde acylhydrazones with methyl iodide in tetrahydrofuran. The reaction proceeds with the exclusive formation of the N-methyl derivative without any concurrent O-methylation side reactions. In addition, the N-methylation reaction occurred simultaneously with a complete deprotection step (elimination of the metal ion). As a result, the N-methyl product was obtained in excellent purity without time-consuming chromatographic workup. A free N-2-hydroxybenzaldehyde acylhydrazone ligand could not be methylated under the same conditions.

 Please wait while we load your content...
Please wait while we load your content...