Imidazolium-based ionic liquids with different fatty acid anions: phase behavior, electronic structure and ionic conductivity investigation†
Abstract
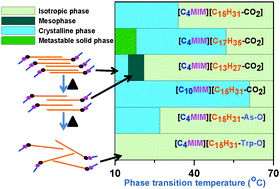
The thermal phase behaviors of a series of newly designed 1-alkyl-3-methylimidazolium ionic liquids (ILs) of different chain length fatty acid carboxylate anions are investigated. The length of the alkyl chain of the carboxylate anion in IL influences the phase transition temperature of their crystalline solid phase and the mesophase stability. When the palmitate anion of the IL is replaced with palmitoyl ascorbate and palmitoyl-L-tryptophanate anions, its melting temperature decreases and eventually vanishes. The influence of structural modulation of ILs on their ionic conductivities is also studied. The interaction between the 1-alkyl-3-methylimidazolium cation and the fatty acid carboxylate anion is established by using ab initio based DFT calculations. The associated energies for single ion pair formation of these ILs are computed and are successfully correlated with the experimental findings, which finally leads to the most reasonable arrangement of the IL molecules in different phases.

 Please wait while we load your content...
Please wait while we load your content...