Solar thermal harvesting for enhanced photocatalytic reactions
Abstract
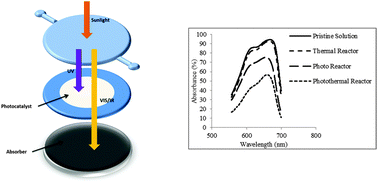
The Shockley–Queisser limit predicts a maximum efficiency of 30% for single junction photovoltaic (PV) cells. The rest of the solar energy is lost as heat and due to phenomena such as reflection and transmission through the PV and charge carrier recombination. In the case of photocatalysis, this maximum value is smaller since the charge carriers should be transferred to acceptor molecules rather than conductive electrodes. With this perspective, we realize that at least 70% of the solar energy is available to be converted into heat. This is specifically useful for photocatalysis, since heat can provide more kinetic energy to the reactants and increase the number of energetic collisions leading to the breakage of chemical bonds. Even in natural photosynthesis, at the most 6% of the solar spectrum is used to produce sugar and the rest of the absorbed photons are converted into heat in a process called transpiration. The role of this heating component is often overlooked; in this paper, we demonstrate a coupled system of solar thermal and photocatalytic decontamination of water by titania, the most widely used photocatalyst for various photo reactions. The enhancement of this photothermal process over solely photocatalytic water decontamination is demonstrated to be 82% at 1× sun. Our findings suggest that the combination of solar thermal energy capture with photocatalysis is a suitable strategy to utilize more of the solar spectrum and improve the overall performance.

 Please wait while we load your content...
Please wait while we load your content...