Probing the photochemistry of chemisorbed oxygen on TiO2(110) with Kr and other co-adsorbates†
Abstract
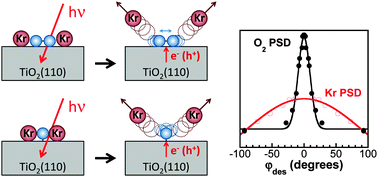
Weakly-bound atoms and molecules (Ar, Kr, Xe, CO, CH4, CO2, CH3OH, N2O, and N2) are used to probe the photochemical interactions of chemisorbed oxygen on rutile TiO2(110). Ultraviolet irradiation of chemisorbed oxygen co-adsorbed with the probe species leads to photon-stimulated desorption (PSD) of some of the probe species (e.g. Kr and CH4), but not others (e.g. CO2 and N2O). Without chemisorbed oxygen, the PSD yields of all the probe species are very low or not observed. Surprisingly, both chemisorbed O2 and oxygen adatoms, Oa, are photo-active for desorption of Kr and other weakly-bound species. To our knowledge, this is the first evidence for photo-activity of Oa on TiO2(110). The Kr PSD yield increases with increasing coverage of Kr and of chemisorbed oxygen. For Kr, the angular distribution of the photodesorbed atoms is approximately cosine. The Kr distribution is quite different from the angular distribution for the O2 PSD, which is sharply peaked along the surface normal. We propose that various forms of chemisorbed oxygen are excited by reactions with electrons and/or holes created in the TiO2 substrate by UV photon irradiation. The photo-excited oxygen collides with, and transfers energy to, neighboring co-adsorbed atoms or molecules. For co-adsorbates with a small enough binding energy to the substrate, desorption may result. The observed phenomenon provides a new tool for studying photochemical processes.

 Please wait while we load your content...
Please wait while we load your content...