Exploring the coordination chemistry of bifunctional organoarsonate ligands: syntheses and characterisation of coordination polymers that contain 4-(1,2,4-triazol-4-yl)phenylarsonic acid†
Abstract
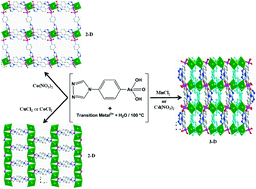
This account describes the coordination chemistry of a novel bifunctional arsonic acid ligand, 4-(1,2,4-triazol-4-yl)phenylarsonic acid (H2TPAA) that contains a triazole group. Hydrothermal reactions of transition metal salts with H2TPAA produced five unprecedented coordination polymers: [Co(H2TPAA)(HTPAA)2]·H2O (1), M(HTPAA)Cl·2H2O (M = Cu (2), Co (3)) and M(HTPAA)2 (M = Mn (4), Cd (5)). These five polymers have been fully characterized by single crystal and powder X-ray diffraction, thermogravimetric analysis, infra-red spectroscopy and elemental analysis. Single crystal X-ray diffraction reveals that 1, 2 and 3 adopt 2-D layered structural motifs whereas 4 and 5 are 3-D frameworks. 1 and 3 are the first examples of arsonate-stabilised cobalt(II) coordination polymers. Likewise, 4 is recognised as the first record of a MnII arsonate coordination polymer with 3-D framework topology. Its isostructural Cd analogue 5 shows characteristic ligand-centered fluorescence properties. Magnetic measurements of 2 and 4 reveal predominant antiferromagnetic interactions between their respective spin centers while, in agreement with its chain structure, 3 exhibits one-dimensional magnetic behavior with weak ferromagnetic interactions between the spin centres.

 Please wait while we load your content...
Please wait while we load your content...