Syntheses, structures, luminescence and molecular recognition properties of four new cadmium carboxyphosphonates with 2D layered and 3D supramolecular structures†
Abstract
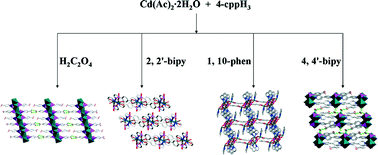
Four new cadmium carboxyphosphonates with 2D layered and 3D supramolecular structures, namely, Cd3[(4-cppH)2(4-cppH2)2] (1), Cd[(2,2′-bipy)(4-cppH)] (2), [Cd3(1,10-phen)3(4-cpp)2]·6H2O (3) and [Cd(4,4′-bipy)(4-cppH)(H2O)2]·2H2O (4) (4-cppH3 = 4-carboxyphenylphosphonic acid, 2,2′-bipy = 2,2′-bipyridine, 1,10-phen = 1,10-phenanthroline and 4,4′-bipy = 4,4′-bipyridine), were synthesized under hydrothermal conditions. In compound 1, {Cd(1)O6}, {Cd(2)O6}, {Cd(3)O6} and {CPO3} polyhedra form a layer in the ab plane via edge- and corner-sharing. Neighboring layers assemble into a 3D supramolecular network by hydrogen bonding interactions. The structure of compound 2 shows a new layered structure, in which the interconnection of two {Cd(1)O4N2} and two {CPO3} polyhedra via corner-sharing forms a tetranuclear cluster, and the so-built tetranuclear clusters are bridged by phosphonate ligands into a 2D layer. For compound 3, {Cd(1)O4N2}, {Cd(2)O4N2}, {Cd(3)O3N2} and {CPO3} polyhedra are interconnected by carboxyphosphonate ligands to a 2D layer in the ac plane. Then the adjacent layers are further assembled into a 3D supramolecular structure through π–π stacking interactions. Cd(II) ions in compound 4 are bridged by 4,4′-bipy molecules into layers in the ab plane. These layers are held together by hydrogen bonds into a 3D supramolecular structure. The thermal stabilities and luminescence properties of compounds 1–4 were investigated. Interestingly, compound 1 is selective and reversible for sensing of DMF and acetone.

 Please wait while we load your content...
Please wait while we load your content...