Combinative method using multi-components quantitation by single reference standard and HPLC fingerprint for comprehensive evaluation of Rhodiola crenulata H.Ohba†
Abstract
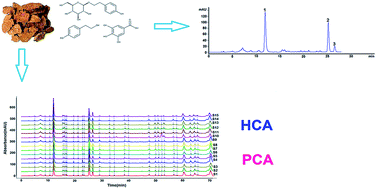
A combinative method using HPLC-DAD quantitative analysis and fingerprints were developed to evaluate the quality of Rhodiola crenulata H.Ohba (R. crenulata) from different origins. In this study, three phenolic compounds were first simultaneously determined by the single standard to determine multi-components (SSDMC) method. The chromatographic separation was performed on a Merck Purospher STAR RP-18 column (4.6 × 250 mm, 5 μm) with a gradient-elution program within 28 min at 275 nm wavelength. The method has achieved desired specificity, linearity (r2 ≥ 0.9999), precision, accuracy (95–105%), stability and robustness. Compared with the normal external standard method (ESM), this alternative method can be used for simultaneous determination of multiple indices effectively and accurately, and can resolve the problem of inadequate chemical standards. Moreover, the herbal chromatographic fingerprint was calculated using a similarity evaluation system and SPSS 19.0 software as a result of analysing all of the R. crenulata samples. The obtained data showed good repeatability and stability of the chromatographic fingerprint. The 15 R. crenulata samples from different origins were classified by hierarchical clustering analysis (HCA) and principal components analysis (PCA) according to the characteristic of common peaks. Moreover, similarity values were all more than 0.90. This study demonstrated that a combination of the chromatographic quantitative analysis and fingerprint offers an efficient way to quality consistency evaluation of R. crenulata.

 Please wait while we load your content...
Please wait while we load your content...