Supramolecular enhancement of aggregation-induced emission and its application in cancer cell imaging†
Abstract
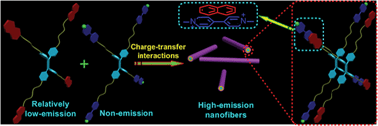
Compared with conventional fluorophores which are often quenched in the aggregate state or at high concentration due to concentration quenching or aggregation-caused quenching, tetraphenylethene (TPE)-based organic fluorogens exhibit an extraordinary aggregation-induced emission (AIE) feature, providing a new platform for the development of fluorescence light-up molecules and photostable nanoaggregates for specific analyte detection and imaging. However, self-assembly of TPE-based building blocks can hardly be achieved without introduction of other driving forces due to the propeller-shaped structure and the dynamic rotation of the phenyl rings of the TPE unit. Herein, two four-armed TPE derivatives containing electron-rich naphthalene (TPE-NP) and electron-deficient paraquat (TPE-PQ) groups, respectively, were designed and synthesized. Driven by charge-transfer (CT) interactions, a complex formed between TPE-NP and TPE-PQ. It self-assembled into nanorods in a 1D packing mode, resulting in the restriction of intramolecular rotation to enhance the AIE effect significantly. A difunctional negatively charged water-soluble pillar[6]arene (H) was used to reduce the toxicity of TPE-PQ by forming a stable inclusion complex (H4⊃TPE-PQ) with TPE-PQ. A ternary system containing H, TPE-NP and TPE-PQ was utilized as an imaging agent for cancer cells due to the pH-responsiveness of H. Compared to the physiological pH of 7.4, the pH in tumor tissue and endosomes is more acidic, resulting in the disassembly of the host–guest complex H4⊃TPE-PQ and the formation of the AIE-enhanced CT complex between TPE-NP and TPE-NP in cancer cells.

 Please wait while we load your content...
Please wait while we load your content...