Amyloid fibrils as dispersing agents for oligothiophenes: control of photophysical properties through nanoscale templating and flow induced fibril alignment†
Abstract
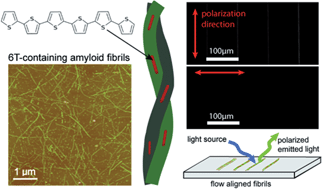
Herein we report that protein fibrils formed from aggregated proteins, so called amyloid fibrils, serve as an excellent dispersing agent for hydrophobic oligothiophenes such as α-sexithiophene (6T). Furthermore, the protein fibrils are capable of orienting 6T along the fibril long axis, as demonstrated by flow-aligned linear dichroism spectroscopy and polarized fluorescence microscopy. The materials are prepared by solid state mixing of 6T with a protein capable of self-assembly. This results in a water soluble composite material that upon heating in aqueous acid undergoes self-assembly into protein fibrils non-covalently functionalized with 6T, with a typical diameter of 5–10 nm and lengths in the micrometre range. The resulting aqueous fibril dispersions are a readily available source of oligothiophenes that can be processed from aqueous solvent, and we demonstrate the fabrication of macroscopic structures consisting of aligned 6T functionalized protein fibrils. Due to the fibril induced ordering of 6T these structures exhibit polarized light emission.

 Please wait while we load your content...
Please wait while we load your content...