The coexisting phase behavior of thermo-responsive copolymer solutions
Abstract
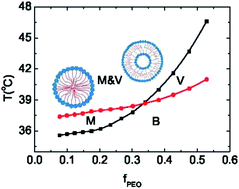
Using a molecular theory for dilute PEO-b-PNIPAm solutions, we first take the formation of hydrogen bonds between copolymer monomers and water molecules into account, which enables us to study the impact of temperature on PEO-b-PNIPAm self-assembly effectively by quantitatively describing the different changes in water affinities of two blocks. With the increase of temperature, hydrogen bonds between PNIPAm and water break down dramatically, resulting in the hydrophobic character of PNIPAm while PEO remains hydrophilic. Amphiphilic copolymers in the aqueous surrounding can aggregate into various structures: micelles and vesicles. According to the equilibrium criterion of the excess grand potential under the conditions of the grand canonical ensemble, we find that both structures are stable and can coexist. Theoretically calculated potentials of mean force of aggregates further verify the coexistence of micelles and vesicles, although the low critical solution temperatures of different aggregates are different under these conditions. A phase diagram as functions of temperature and the weight fraction of PEO (fPEO) is obtained, which shows different regions of micelles, vesicles and their coexistence. It implies the appearance of two types of micelle–vesicle transition: spontaneous and temperature-induced. Since PEO-b-PNIPAm as a thermoresponsive material has a broad range of applications, a systematic investigation of the phase behavior is very useful not only for the scientific interest but also for the practical applications.

 Please wait while we load your content...
Please wait while we load your content...