Production of γ-valerolactone from biomass-derived compounds using formic acid as a hydrogen source over supported metal catalysts in water solvent†
Abstract
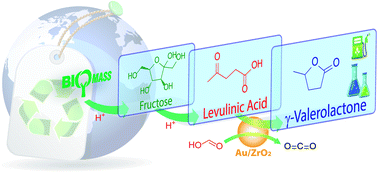
γ-Valerolactone (GVL) is a key intermediate for production of fuels and chemicals. In this research, GVL is synthesized from biomass-derived compounds using formic acid (FA) as a hydrogen source over various supported metal catalysts which are prepared by a simple impregnation or co-precipitation method. Under optimum conditions, levulinic acid (LA) is almost converted to GVL by Ru/C, Ru/SBA, Au/ZrC and Au/ZrO2 catalysts with above 90% yield in water solvent. Especially, the Au/ZrO2 showed excellent activity and recyclability; the Au/ZrO2 catalyst can decompose completely FA to CO2 and H2, which gives high yield of GVL (ca. 97%) from hydrogenation of LA, and can retain its activity for at least 5 recycle runs. GVL is also obtained from one-pot dehydration/hydrogenation reaction of fructose in water solvent. In this reaction, FA plays two roles: an acid catalyst for dehydration of fructose to LA, and a hydrogen source for hydrogenation of the obtained LA over supported metal catalysts. The Au/ZrO2 is the best catalyst for dehydration/hydrogenation reaction with overall GVL yield of 48% and can be reused several times.
- This article is part of the themed collection: Organic chemistry collection

 Please wait while we load your content...
Please wait while we load your content...