One-pot synthesis of pH-sensitive poly(RGD-co-β-amino ester)s for targeted intracellular drug delivery†
Abstract
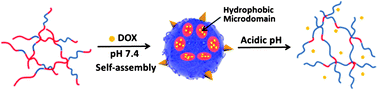
We report a convenient synthetic approach for one-pot preparation of poly(β-amino ester)s copolymerized with peptides. A family of copolymers with tertiary amine groups was synthesized by copolymerizing di(ethylene glycol) diacrylate (DEDA), poly(ethylene glycol) diacrylate (PEGDA), 3-(diethylamino)propylamine (DEPA) and GGRGD peptides using the Michael addition reaction. The hydrophobic/hydrophilic properties of the resulting copolymers are adjusted by altering the feed ratios of DEDA and PEGDA. The copolymers self-assembled into nanoparticles with sizes around 60–140 nm in aqueous media, which were confirmed by dynamic light scattering (DLS) and transmission electron microscopy (TEM) techniques. The copolymers exhibit pH sensitive properties upon introduction of DEPA moieties, which were proved by pyrene fluorescence and pH titration measurements. The acid-triggered dissociation behaviors of the nanoparticles were studied by DLS and Nile red (NR) release experiments, revealing that the sizes of dissociated copolymer nanoparticles were closely relevant to their compositions. The nanoparticles can load the hydrophobic anticancer drug, i.e., doxorubicin (DOX). The DOX-loaded nanoparticles were stable in a neutral phosphate buffer solution with a payload leakage less than 20% at 37 °C. However, a significant acid-triggered DOX release was accomplished at pH 5.0 with release efficiency up to 60–80%. Because of the decoration of Arg-Gly-Asp (RGD) peptides onto the poly(β-amino ester)s, the DOX-encapsulated nanoparticles formed by poly(RGD-co-β-amino ester)s can be internalized by cancer cells via an αvβ3 integrin-mediated endocytosis pathway and accumulated in the lysosomes that provide an acidic environment to promote the release of DOX. Finally, the DOX-encapsulated copolymer nanoparticles with the targeted RGD peptide exhibited higher efficiency to kill U87 human glioblastoma cancer cells than that without RGD, which was further proved by cellular uptake of DOX-loaded nanoparticles.

 Please wait while we load your content...
Please wait while we load your content...