Comparing systemic metabolic responses in mice to single or dual infection with Plasmodium berghei and Heligmosomoides bakeri†
Abstract
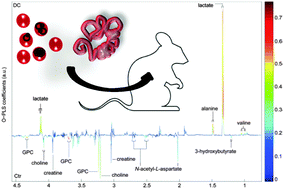
Concomitant infections with Plasmodium and gastrointestinal nematodes are frequently observed in humans. At the metabolic level, the cross-talk between the host and multiple coexisting pathogens is poorly characterized. The purpose of this study was to give a comprehensive insight into the systemic metabolic phenotype of mice with a single or dual infection with Plasmodium berghei and Heligmosomoides bakeri. Four groups of eight NMRI female mice were infected with P. berghei or H. bakeri, or with both species concurrently. An additional group remained uninfected, and served as control. Mice were sacrificed at day 19 of the experiment. We collected samples from the liver, spleen, kidney, three intestinal regions, and four brain regions. All biological samples were subjected to 1H nuclear magnetic resonance spectroscopy, combined with multivariate data analysis, to establish metabolic fingerprints of each tissue from the various infection groups. Compared to uninfected mice, single and dual species infection models showed unique metabolic profiles. P. berghei exerted major effects on glycolysis, tricarboxylic acid cycle, and nucleotide and amino acid metabolism in all studied tissues with the exception of the gut. H. bakeri was characterized by a dysregulation of choline and lipid metabolism in most tissues examined with a particularly strong imprint in the jejunum. Simultaneous co-infection with P. berghei and H. bakeri induced the strongest and most diverse effects in the liver and spleen but led to only minor changes in the intestinal and cerebral parts assessed. Infection with P. berghei showed more pronounced and systemic alterations in the mice metabolic profile than H. bakeri infection. The metabolic fingerprints in the co-infection models were driven by P. berghei infection, whilst the presence of H. bakeri in co-infections had little effect. However, simultaneous co-infection showed indeed the least metabolic disruptions in the peripheral tissues, namely the gut and brain.

 Please wait while we load your content...
Please wait while we load your content...