A three-dimensional neuronal culture technique that controls the direction of neurite elongation and the position of soma to mimic the layered structure of the brain†
Abstract
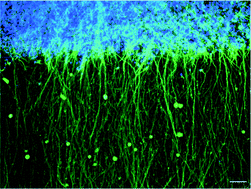
Reconstruction techniques can mimic tissue structure using three-dimensional (3D) substrates or scaffolds to facilitate functional tissue engineering for transplantation or robust experimental models. Neuron-based tissue engineering is being developed to treat neuronal diseases and to understand neuronal function. However, few 3D neuronal tissue reconstruction techniques are available because structural control is hindered by the complexity and polarity of neurites. In this study, we developed a 3D reconstruction neuronal tissue technique using collagen fiber orientation and polydimethylsiloxane microchambers. This technique mimicked the layered structure of the brain (cerebral cortex) on a chip. We used this method to produce 3D neuronal networks by controlling (1) the position of somata and (2) the direction of neurite elongation in the 3D space. The somata area comprised a three-cell layer, and the cell density was equivalent to living tissue. Intracellular Ca2+ imaging and extracellular recordings using multielectrode arrays chip detected interlayer synchronous firings in a 3D reconstructed neuronal network. We confirmed that the interlayer propagation was chemical synaptic transmission by pharmacological experiments and that the velocity of propagation was equivalent to biological tissue. Furthermore, we demonstrated the reconstruction of 3D neuronal networks using neurons derived from human induced pluripotent stem cells. This 3D neuronal culture technique could be a useful tool for regenerative medicine and a drug screening model.

 Please wait while we load your content...
Please wait while we load your content...