Enhanced photocatalytic activity and stability of semiconductor by Ag doping and simultaneous deposition: the case of CdS†
Abstract
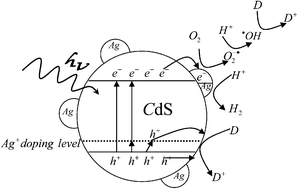
Herein, it is demonstrated how the photocatalytic activity and stability of a semiconductor can be enhanced through metal doping and simultaneous deposition in the case of CdS. The CdS which is doped and simultaneously deposited by Ag (denoted as Ag–CdS thereafter) is successfully prepared by a one-step hydrothermal method. The prepared Ag–CdS samples have enhanced photocatalytic activity under simulated sunlight compared with pure CdS and Ag-deposited CdS for organic pollutant degradation and H2 production from water splitting. The main reason for the enhanced activity is due to the fast transfer of photogenerated electrons from the conduction band of CdS to Ag nanoparticles on the surface, which suppresses the recombination of photogenerated electrons and holes. As for the enhanced stability of the Ag–CdS sample, the doping with Ag creates an acceptor energy level near the valence band of CdS. The holes will inject into the acceptor energy level freely, leading to the weak oxidative ability of holes in the excited semiconductor. Moreover, the more stable and more easily formed chemical bond between Ag+ substituted for Cd2+ and S2+ makes the sample more stable than Ag-deposited CdS. The Ag–CdS photocatalyst with high activity and stability can be used in future applications.

 Please wait while we load your content...
Please wait while we load your content...