Coenzyme-inspired chemistry 2: 4,5-dihydroimidazolium ylides (NHCs) and the reactions of 2-(1-hydroxyalkyl)-4,5-dihydroimidazoles
Abstract
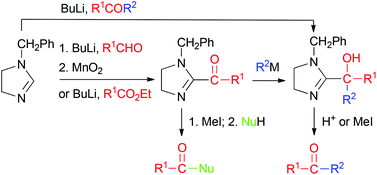
Ketones are prepared from aldehydes via 1-benzyl-2-(1-hydroxyalkyl)-4,5-dihydroimidazoles (adducts of the aldehydes with 1-benzyl-2-lithio-4,5-dihydroimidazoles) whereas 1-benzyl-2-(1-oxoalkyl)-4,5-dihydroimidazoles are shown to act as acyl transfer reagents via C–C bond cleavage. 4,5-Dihydroimidazolium ylides (NHCs) are intermediates in both processes, which constitute thiamine-inspired C–C bond formation and cleavage protocols.

 Please wait while we load your content...
Please wait while we load your content...