A model of membrane contraction predicting initiation and completion of bacterial cell division
Abstract
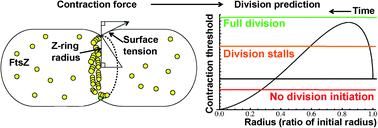
Bacterial cell division involves a complex and dynamic sequence of events whereby
We are excited to let you know that our journals content will be migrating to the Silverchair platform, with a planned launch in summer 2026.
All of our content will still be hosted at pubs.rsc.org and the new platform will provide a more intuitive reading and navigation experience, along with improved discovery and indexing of your work.
Details on the move can be found in our partnership announcement.
* Corresponding authors
a
Molecular Organisation and Assembly in Cells Doctoral Training Centre, Senate House, University of Warwick, Coventry, UK
E-mail:
C.E.Dow@warwick.ac.uk
b Department of Chemistry and Warwick Centre for Analytical Science, University of Warwick, Coventry, UK
c School of Life Sciences, University of Warwick, Coventry, UK
d Mathematics Institute, University of Warwick, Coventry, UK
Bacterial cell division involves a complex and dynamic sequence of events whereby

 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?
 Fetching data from CrossRef.
Fetching data from CrossRef.
This may take some time to load.
Loading related content
