Towards efficient solar hydrogen production by intercalated carbon nitride photocatalyst†
Abstract
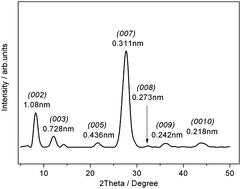
The development of efficient photocatalytic material for converting solar energy to hydrogen energy as viable alternatives to fossil-fuel technologies is expected to revolutionize energy shortage and environment issues. However, to date, the low quantum yield for solar hydrogen production over photocatalysts has hindered advances in the practical applications of photocatalysis. Here, we show that a carbon nitride intercalation compound (CNIC) synthesized by a simple molten salt route is an efficient polymer photocatalyst with a high quantum yield. We found that coordinating the alkali metals into the C–N plane of carbon nitride will induce the un-uniform spatial charge distribution. The electrons are confined in the intercalated region while the holes are in the far intercalated region, which promoted efficient separation of photogenerated carriers. The donor-type alkali metal ions coordinating into the nitrogen pots of carbon nitrides increase the free carrier concentration and lead to the formation of novel nonradiative paths. This should favor improved transport of the photogenerated electron and hole and decrease the electron–hole recombination rate. As a result, the CNIC exhibits a quantum yield as high as 21.2% under 420 nm light irradiation for solar hydrogen production. Such high quantum yield opens up new opportunities for using cheap semiconducting polymers as energy transducers.

 Please wait while we load your content...
Please wait while we load your content...