A computational study of acid catalyzed aerosol reactions of atmospherically relevant epoxides†
Abstract
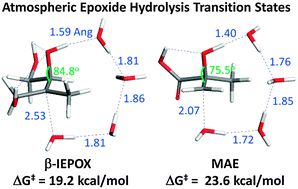
Epoxides are important intermediates of atmospheric isoprene oxidation. Their subsequent reactions in the particle phase lead to the production of organic compounds detected in ambient aerosols. We apply density functional theory to determine the important kinetic factors that drive epoxide reactions in the particle phase. Specifically, the importance of acid catalysis and solvent polarity are investigated using a variety of epoxides and nucleophiles. The condensed phase is modeled using molecular clusters immersed in a dielectric continuum and a majority of the calculations are performed with the M062x density functional and the 6-311++G** basis set. Calculations of acid catalyzed epoxide hydrolysis transition states for simple primary, secondary and tertiary epoxides are consistent with an A-2 mechanism where the nucleophile (water) interacts with an epoxide carbon in the transition state. By applying transition state theory to this mechanism, the overall rate constants of epoxide reactions such as hydrolysis, organosulfate formation, organonitrate formation and oligomerization are determined. The calculations indicate that the acid catalyzed hydrolysis rate constant of 2-methyl-2,3-epoxybutane-1,4-diol (β-IEPOX – an isoprene epoxide produced under low NOx conditions) is approximately 30 times greater than 2-methyl-2,3-epoxypropanoic acid (MAE – methacrylic acid epoxide derived from isoprene and produced at high NOx concentrations). Furthermore, acid catalyzed organosulfate formation and epoxide oligomerization reactions are competitive and appear to be kinetically favorable over the hydrolysis of IEPOX.

 Please wait while we load your content...
Please wait while we load your content...