A highly selective and sensitive fluorescence ratiometric probe for cyanide and its application for the detection of cyanide in natural water and biological samples†
Abstract
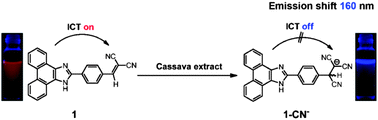
A fluorescent ratiometric probe, compound 1, for cyanide has been rationally constructed based on an intramolecular charge transfer (ICT) mechanism. Upon treatment with cyanide, probe 1 exhibited a fluorescence ratiometric response, with the emission wavelength displaying a very large shift (up to 160 nm). When 2 equivalents of cyanide were added, the emission ratios (I458/I618) of probe 1 changed dramatically from 0.027 to 28.306. In addition, the emission ratios (I458/I618) showed good linearity with the cyanide concentration in the range of 0–6.0 μM, and the detection limit measured to be 0.126 μM. The NMR experiments and theoretical calculations confirmed that the ratiometric response of probe 1 to cyanide is due to the nucleophilic addition reaction of the cyanide with the vinyl malononitrile group of probe 1, which results in the inhibition of the ICT process in the probe. Importantly, probe 1 has been applied to detection of cyanide in a natural water sample with satisfactory recovery. Furthermore, the quantitative determination of endogenous cyanide in cassava root demonstrated that the probe can be employed to detect endogenous cyanide in real biological samples.

 Please wait while we load your content...
Please wait while we load your content...