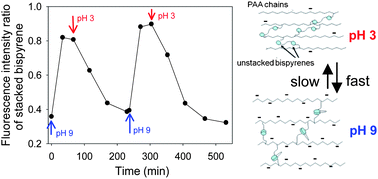
Self-construction of films using polyelectrolytes bearing complementary chemical groups is a recent emerging concept in the coating field. Herein, we describe the one-pot morphogen film self-construction through formation of covalent bonds between polyelectrolytes and homobifunctional spacers by Huisgen click-chemistry. One-pot solution of poly(acrylic acid) bearing alkyne groups (PAA-Alk), bisazides ethylene glycol (Az-EGn-Az) spacers and CuSO4 was brought into contact with a gold electrode. Cu(I), the morphogen, was generated electrochemically from Cu(II) at the electrode/one-pot solution interface. The PAA-Alk/Az-EGn-Az film buildup was investigated by varying the length of the EGn spacer (n = 2, 13 and 50) leading to different growth rates. By increasing the pH of the solution, a reversible swelling of the films (up to 400% for n = 13 or 50) is obtained due to electrostatic repulsion between PAA chains. Further investigations on film swelling were performed by using PAA-Alk and an original bisazide spacer based on two pyrene units connected by a triamine spacer (Az-bispyrene-Az). This spacer has the property to adopt an extended conformation under acidic conditions and a “stacked” conformation under basic conditions, which both can be monitored by fluorimetry. Despite the mechanical tension due to film swelling at basic pH, bispyrene spacers adopt a stacked conformation. This allowed us to monitor the pH within PAA-Alk/Az-bispyrene-Az films. The pH remains fairly stable and below the pKa of PAA when the pH of the contacting solution varies from pH 5 to pH 7 and increases above pH 8, which explains why film swelling takes place essentially above pH 8.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...