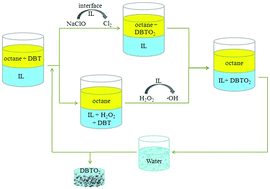
Room-temperature ionic liquids (ILs) are commonly used for desulfurization by extraction for their unique solvent characteristics. In this study, 1-ethyl-3-(4-sulfobutyl)imidazolium bis(trifluoromethanesulfonyl)imide ([EimC4SO3H]NTf2) was used as catalyst and extractant for desulfurization of DBT in n-octane. The results show that dibenzothiophene (DBT) could be removed from the oil phase effectively by this process, and that the sulfur content in a model oil could be decreased from 1600 ppm to less than 20 ppm. The extraction efficiency of the IL was greatly influenced by the structure of the anions and cations. The order of extraction capability of ILs was [EimC4SO3H]NTf2 > [EimC4SO3H]HSO4 > [C4Py]NTf2 > [Bmim]NTf2 > [Bmim]PF6. Experiments on the recycling and reuse of the room-temperature IL were also performed, and we found that [EimC4SO3H]NTf2 could be recycled 5 times without significant decrease in the desulfurization activity. The extraction mechanism was also probed in detail, and we found that DBT was oxidized to its corresponding sulfone by H2O2 and NaClO, which was then extracted by the IL phase. However, different extraction mechanisms are at work with different oxidants.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...