Pressure dependent mechanistic branching in the formation pathways of secondary organic aerosol from cyclic-alkene gas-phase ozonolysis†
Abstract
The gas-phase ozonolysis of cyclic-
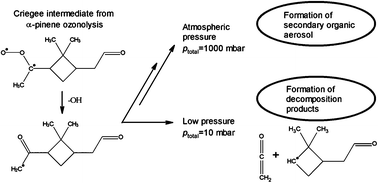
- This article is part of the themed collection: Leuven gas kinetics meeting

 Please wait while we load your content...
Please wait while we load your content...