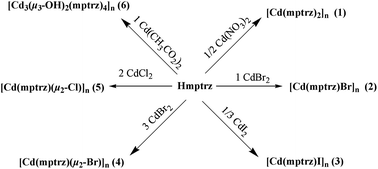
Hydro(solvo)thermal reactions of cadmium(II) salts with 4-methyl-1,2,4-triazole-3-thiol (Hmptrz) afforded six novel cadmium(II) coordination polymers, namely, [Cd(mptrz)2]n (1), [Cd(mptrz)X]n (X = Br (2) and I (3)), [Cd(mptrz)(μ2-X)]n (X = Br (4) and Cl (5)), and [Cd3(μ3-OH)2(mptrz)4]n (6). Compound 1 is a 1D looped chain structure. Compounds 2 and 3 are isostructural and have 1D ladder structures. Compounds 4 and 5 are isomorphs and feature 2D layered structures. Cadmium(II) bromide formed 1D ladder of 2 and 2D layer of 4, representing an unusual example of Br bond isomerism. Compound 6 has a 1D tape structure with an uncommon 1D inorganic ⋯Cd2(μ3-OH)2–Cd–Cd2(μ3-OH)2⋯ chain. The structural diversity of these materials is reflected in the variety of coordination polyhedra displayed by the cadmium sites: tetrahedral; trigonal bipyramidal; octahedral. The solid state photoluminescence of all compounds have been investigated. However, only compound 6 exhibits photoluminescence with an emission maximum at 435 nm upon excitation at 346 nm. Solid-state density functional theory (DFT) calculations were performed on compound 6. In addition, these compounds were characterized by IR, elemental analysis, and thermogravimetric analysis.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...