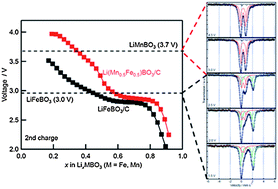
The lithium-ion battery is the most advanced energy storage system that has conquered the portable electronics market, but it has been considered unsuitable for large-scale applications such as plug-in hybrid vehicles due to cost, safety, and calendar life issues. Research in materials science is approaching a possible solution to these problems by the combination of abundant Fe as a redox center and covalent (PO4)3−oxyanion units with fixed oxygen, leading to the use of LiFePO4 with olivine-type structure as a cathode material. A high level of stability, safety, significant cost reduction, and huge power generation are now on the verge of being guaranteed. Replacing the (PO4)3− anion to smaller and lighter (BO3)3−, namely LiMBO3 (M = transition metal) compounds, could possibly increase the theoretical capacity by approximately >1.3 times; however, such systems have not been thoroughly investigated to date. Here we present the successful solid state synthesis of a complete solid solution of Li(MnxFe1−x)BO3 with monoclinic structure and the optimization processes to maximize its performance as a lithium battery cathode. Details of the intrinsic electrode properties, including the phase stability issues as well as degradation mode, are demonstrated, followed by systematic investigation of the reaction mechanisms by ex situX-ray diffraction, Mössbauer spectroscopy, X-ray absorption spectroscopy, and ab initio calculations.

 Please wait while we load your content...
Please wait while we load your content...