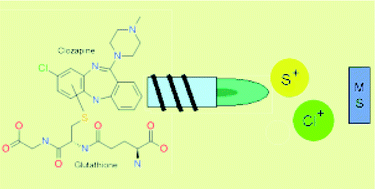
In this study, a novel method was developed for tracing down reactive drug metabolites, the formation of which in the human body constitutes an important health risk as a result of their capability to bind to body proteins and DNA. Clozapine was used as a model because this drug forms both reactive and stable metabolites. Glutathione, which forms complexes with reactive metabolites, was added in order to trap reactive species of clozapine, formed by degradation in an electrochemical cell, thereby mimicking the real drug metabolism process. The method developed is based on the use of reversed-phase HPLC as a chromatographic separation technique and inductively coupled plasma-mass spectrometry (ICP-MS) for monitoring of Cl and S. Via Cl-monitoring, all metabolites of the Cl-containing clozapine can be detected, whereas S-monitoring allows for the detection of the S-containing molecule glutathione and its conjugates with reactive metabolites. The spectral overlap of the signals of 32S+ and 34S+ with those of 16O16O+ and 16O18O+, respectively, was tackled in 2 ways. On one hand, a quadrupole-based ICP-MS instrument, equipped with a dynamic reaction cell, was used. O2 was used as a reaction gas to convert the S+ ions to a sufficient extent into the corresponding SO+ species. This did not yield optimal results, due to pronounced ArC+ signals at mass 48 and 50 upon introduction of methanol into the ICP. On the other hand, a sector-field ICP-MS instrument operated at medium mass resolution permitted interference-free monitoring of the S+-signals. A new type of skimmer cone – termed X-skimmer – was evaluated and its use resulted in a 4-fold increase in the sensitivity in a methanolic environment, providing a limit of detection of 1 μg L−1 for S. The chromatograms obtained via HPLC-SF-ICP-MS permitted differentiation between reactive and stable metabolites. As a result, the method developed looks very promising for the detection of glutathione conjugates and shows potential for their quantification in early stages of drug development when a radiolabeled compound is not yet available.

 Please wait while we load your content...
Please wait while we load your content...