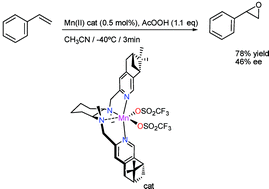
A novel family of chiral manganese complexes Λ-1(CF3SO3) and Δ-2(CF3SO3), have been stereoselectively prepared, characterized and studied as epoxidation catalysts. The complexes are structurally related to [MnII(CF3SO3)2(α-MCP)] (MCP = N,N′-dimethyl-N,N′-bis(2-pyridylmethyl)cyclohexane-trans-1,2-diamine), recently reported as a very efficient epoxidation catalyst in combination with peracetic acid. Pinene rings have been fused to the 4 and 5 positions of the two pyridine groups of the ligand, giving rise to complexes where the two labile binding sites of the manganese ion are confined in a better-defined chiral pocket than in the parent [MnII(CF3SO3)2(α-MCP)]. Chirality in these complexes arises from the stereochemistry of the trans-diaminocyclohexane ring, from the pinene ring and also from the topological chirality adopted by the ligand upon binding to the manganese ion. While previous studies have demonstrated that small modifications in the structure of the MCP ligand result in a dramatic loss of efficiency, Λ-1(CF3SO3) and Δ-2(CF3SO3) exhibit comparable catalytic activity to [MnII(CF3SO3)2(α-MCP)]. In addition, the complexes exhibit a remarkable stereoselectivity (up to 46% ee) in the epoxidation of selected substrates. The results reported in this work point towards modification of the 4 and 5 positions of the pyridine groups as a new strategy towards the design of stereoselective versions of this family of highly active and environmentally benign epoxidation catalysts.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...