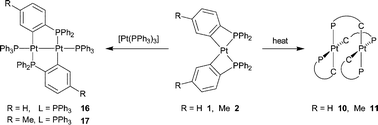
When the ortho-metallated complexes cis-[Pt(κ2-C6H3-5-R-2-PPh2)2] (R = H 1, Me 2) are either heated in toluene or treated with CO at room temperature, one of the four-membered chelate rings is opened irreversibly to give dinuclear isomers [Pt2(κ2-C6H3-5-R-2-PPh2)2(µ-C6H3-5-R-2-PPh2)2] (R = H 10, Me 11). A single-crystal X-ray diffraction study shows the Pt⋯Pt separation in 10 to be 3.3875(4) Å. By-products of the reactions of 1 and 2 with CO are polymeric isomers (R = H 13, Me 14) in which one of the P–C ligands is believed to bridge adjacent platinum atoms intermolecularly. In contrast to the behaviour of 1 and 2, when cis-[Pt(κ2-C6H3-6-Me-2-PPh2)2] (cis-3) is heated in toluene, the main product is trans-3, and reaction of cis-3 with CO gives a carbonyl complex [Pt(CO)(κ1-C-C6H3-6-Me-2-PPh2)(κ2-C6H3-6-Me-2-PPh2)] 15, in which one of the carbanions is coordinated only through the carbon. Formation of a dimer analogous to 10 or 11 is sterically hindered by the 6-methyl substituent. Comproportionation of 1 or 2 with [Pt(PPh3)2L] (L = PPh3, C2H4) gives diplatinum(I) complexes [Pt2(µ-C6H3-5-R-2-PPh2)2(PPh3)2] (R = H 16, Me 17). An X-ray diffraction study shows that 17 contains a pair of planar-coordinated metal atoms separated by 2.61762(16) Å. There is no evidence for the formation of an analogue containing µ-C6H3-6-Me-2-PPh2. The axial PPh3 ligands of 16 are readily replaced by ButNC giving [Pt2(µ-2-C6H4PPh2)2(CNBut)2] 18, which is protonated by HBF4 to form a µ-hydridodiplatinum(II) salt [Pt2(µ-H)(µ-2-C6H4PPh2)2(CNBut)2]BF4 [21]BF4. The JPtPt values in [21]BF4 and 18, 2700 Hz and 4421 Hz, respectively, reflect the weakening of the Pt–Pt interaction caused by protonation. Similarly, 16 and 17 react with the electrophiles iodine and strong acids to give salts of general formula [Pt2(µ-Z)(µ-C6H3-5-R-2-PPh2)2(PPh3)2]Y (Y = Z = I, R = H 19+, Me 20+; Z = H, Y = BF4, PF6, OTf, R = H 22+; Z = H, Y = PF6, R = Me 23+). A single-crystal X-ray diffraction study of [23]PF6 shows that the cation has an approximately A-frame geometry, with a Pt⋯Pt separation of 2.7888(3) Å and a Pt–H bond length of 1.62(1) Å, and that the 5-methyl substituents have undergone partial exchange with the 4-hydrogen atoms of the PPh2 groups of the bridging carbanion. The latter observation indicates that the added proton of [23]+ undergoes a reversible reductive elimination–oxidative addition sequence with the Pt–C(aryl) bonds.

 Please wait while we load your content...
Please wait while we load your content...