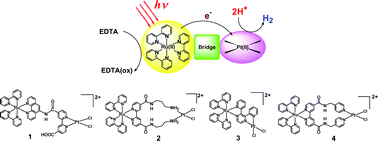
The photo-hydrogen-evolving activity (activity to enhance the photochemical EDTA-reduction of water into molecular hydrogen) was evaluated for three different Ru(II)Pt(II) dimers with a general formula of [(bpy)2Ru(µ-bridge)PtCl2]2+
(bpy = 2,2′-bipyridine; bridge = 4,4′-bis(N-(3-aminopropyl)carbamoyl)-2,2′-bipyridine (L1), 2,3-bis(2-pyridyl)pyrazine (L2), and 4,4′-bis(N-(4-pyridyl)methylcarbamoyl)-2,2′-bipyridine (L3); EDTA = ethylenediaminetetraacetic acid disodium salt). A new Ru(II)Pt(II) complex, [(bpy)2Ru(µ-L3)PtCl2]2+, was synthesized and characterized. It was confirmed that all three compounds are ineffective towards photochemical H2 production. In each case, an acetate-buffer solution (pH = 5) containing the Ru(II)Pt(II) dimer and EDTA was photolysed using a 350-W Xe lamp under an Ar atmosphere, during which the amount of H2 evolved was analysed by gas chromatography. Additional photolysis experiments were carried out by adding [Ru(bpy)3]2+ and methylviologen (N,N′-dimethyl-4,4′-bipyridinium) to the photolysis solutions described above to test the H2-evolving activity of the Pt(II) unit involved in these Ru(II)Pt(II) dimers. As a result, the Pt(II) units involved in the L1 and L2 compounds were found to be active as an H2-evolving catalyst, while that of the L3 compound was found to show no activity at all. The extent of intramolecular electron-transfer quenching from the 3MLCT excited state of the [Ru(bpy)3]2+ derivative to the tethering Pt(II) catalyst centre was investigated by comparison of the luminescence spectra of these compounds, together with the related compounds. The results showed that the quenching of the 3MLCT luminescence is not at all enhanced in either the L1 or the L3 compounds. On the other hand, the L2 compound is strongly quenched as previously reported. In addition to the above studies, the H2-evolving activity of some Pt(II) monomers, cis-PtCl2(NH3)2, PtCl2(en)
(en = ethylenediamine), cis-PtCl2(4-methylpyridine)2, PtCl2(2,2′-bipyrimidine), PtCl2(4,4′-dicarboxy-2,2′-bipyridine), and [PtCl(terpy)]+
(terpy = 2,2′:6′,2″-terpyridine), were similarly investigated in the presence of EDTA, [Ru(bpy)3]2+ and methylviologen, since they were regarded as structural analogues of the Pt(II) units involved in the L1–L3 compounds. The compounds having a cis-Pt(II)Cl2 unit were generally found to show high H2-evolving activity. This was interpreted in terms of the ligation of negatively charged chloride anions leading to the destabilization of the Pt(II) dz2orbital responsible for the hydrogenic activation. Importantly, cis-PtCl2(4-methylpyridine)2 exhibited relatively high activity as an H2-evolving catalyst, suggesting the importance of the flexible rotation of the pyridyl ligands for efficient hydrogenic activation at the axial site of the Pt(II) ion. The DFT calculations also showed the validity of the structure–activity relationship discussed above for the L3 compound.

 Please wait while we load your content...
Please wait while we load your content...