Using spin polarised positive muons for studying guest molecule partitioning in soft matter structures
Abstract
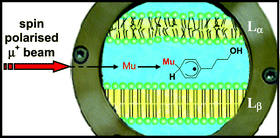
Fully polarised positive muons substituted for protons in organic free radicals can be used as spin labels which reveal information about the structure, dynamics and environment of these radicals. In applications via the technique of avoided-level-crossing muon spin resonance (ALC-μSR), the positive muon has been used to study the partitioning of phenyl alcohols in lamellar phase colloidal dispersions of a cationic dichain surfactant. Here we describe the experimental technique which permits highly sensitive spectroscopy as previously demonstrated for surfactant mixtures. We also demonstrate its capability in the study of partitioning of cosurfactant molecules in surfactant bilayers in order to elucidate the main factors which contribute to cosurfactant ordering at interfaces. The technique takes advantage of the positive muon combining with an electron to a hydrogen-like atom that is called muonium. This atom attaches to a phenyl group, forming a cyclohexadienyl-type radical that contains the muon as a polarised spin label, providing an excellent probe even for very low phenyl alcohol concentrations. The position of one type of resonance, which on the basis of spectroscopic selection rules is denoted as Δ0, is related to the solvent polarity of the radicals’ environment. The results derived from Δ0 measurements reveal a systematic trend where the increasing chain length of the phenyl alcohol results in a deeper immersion of the phenyl ring of the alcohol into the surfactant bilayer with the OH group anchored at the interface. In addition, the data suggest partial penetration of water molecules into the bilayer. Furthermore, data ensuing from a second resonance (called Δ1, which is dependent upon the degree of confinement of the radical within the surfactant aggregate structure) indicates not only that the phenyl alcohol resides in an anisotropic environment, (i.e. that the host molecule is unable to undergo full 3-D reorientation on a timescale of 50 ns), but the resonance line widths indicate that the radicals are undergoing fast rotation about a particular axis, in this instance about the first C–C substituent bond at the phenyl ring. Detailed analysis of these Δ1 line shapes suggests that other types of motion involving reorientation of the above rotation axis are also present. At room temperature, the hydrocarbon chains of the double layers form an aggregate state commonly referred to as the Lβ phase, where the motions of surfactant alkyl chains are effectively frozen out. These chains melt on heating over a temperature range which is solution composition dependent (ca. 51 to 67 °C), but in all cases leading to a liquid-like disordered hydrocarbon regime whilst retaining the overall lamellar structure (and in this state is termed Lα). Above the Lα/Lβ chain ordering phase transition the tracer molecules reside within the bilayer, but below this transition (and depending on their water–oil solubility) they are completely or partly expelled. This interpretation is further supported by Heisenberg spin exchange experiments. The water–bilayer partitioning reflects both typical classical and nonclassical hydrophobic solvation depending on temperature and chain length of phenyl alcohols.

 Please wait while we load your content...
Please wait while we load your content...