The role of halogenated carborane monoanions in olefin hydrogenation catalysed by cationic iridium phosphine complexes†
Abstract
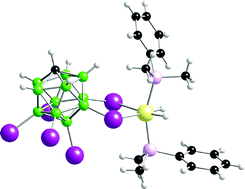
Iridium hydridophosphine complexes of general formula [Ir(PR3)2H2(anion)] (PR3 = PPh3, PMe2Ph; anion = [1-closo-CB11H6Cl6]−, [1-closo-CB11H6I6]−, [BArF4]−) have been prepared by hydrogenation of cyclooctadiene precursor complexes. Solid-state structures of selected examples of these complexes reveal intimate contacts between the carborane anion and cation, with the anion binding through two lower-hemisphere halogen ligands. In CD2Cl2 solution the very weakly coordinating anions [1-closo-CB11H6Cl6]− and [BArF4]− are suggested to favour the formation of solvent complexes such as [Ir(PR3)2H2(solvent)n][anion], while the [1-closo-CB11H6I6]− anion forms a tightly bound complex with the cationic iridium fragment. Calculated ΔG‡ values for anion reorganisation in d8-toluene reflect this difference in interaction between the anions and cation. With the bulky anion [1-closo-CB11Me5I6]− different complexes are formed: Ir(PPh3)H2(1-closo-HCB11Me5I6) and [(PPh3)3Ir(H2)H2][1-closo-HCB11Me5I6] which have been characterised spectroscopically. Diffusion measurements in CD2Cl2 are also consistent with larger, solvent coordinated, complexes for the more weakly coordinating anions and a tighter interaction between anion and cation for [1-closo-CB11H6I6]−. All the complexes show some ion-paring in solution. Comparison with data previously reported for the [1-closo-CB11H6Br6]− anion shows that this anion—as expected—fits between [1-closo-CB11H6Cl6]− and [1-closo-CB11H6I6]− in terms of coordinating ability. Although not coordinating, the large [1-closo-CB11H6Cl6]− and [BArF4]− anions do provide some stabilisation towards the metal centre, as decomposition to the hydride bridged dimer [Ir2(PPh3)4H5]+ is retarded. This is in contrast to the [PF6]− salt where decomposition is immediate. As expected, complexes with the smaller phosphine PMe2Ph form tighter interactions with the carborane anions. These observations on the interaction between anion and cation in solution are reflected in benchmark hydrogenation studies that show a significant attenuation in rate of hydrogenation of cyclohexane on using the [1-closo-CB11H6I6]− anion or complexes with the PMe2Ph phosphine. We also comment on the reusability of the catalysts and their tolerance to water and oxygen impurities. Overall the catalyst with the [1-closo-CB11H6Br6]− anion shows the best combination of rate of hydrogenation, reusability and tolerance to impurities.

 Please wait while we load your content...
Please wait while we load your content...