Organocatalysis with proline derivatives: improved catalysts for the asymmetric Mannich, nitro-Michael and aldol reactions
Abstract
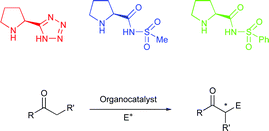
Tetrazole and acylsulfonamide organocatalysts derived from proline have been synthesised and applied to the asymmetric Mannich, nitro-Michael and aldol reactions to give results that are superior to the proline-catalysed counterpart.

 Please wait while we load your content...
Please wait while we load your content...